Beta Hydroxybutyrate – What Is It? What Role Does It Play For Stressed Hearts?
Beta hydroxybutyrate, a ketone body, has been linked to the failing heart's energy metabolism.
Author:Suleman ShahReviewer:Han JuJul 18, 20221 Shares267 Views
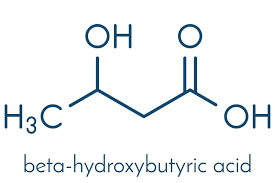
A significant change in energy metabolism is one of the markers of a failing human heart.
Beta-hydroxybutyrate, a ketone body, has been linked to the failing heart's energy metabolism.
Beta-hydroxybutyrate increases in the failing heart, acting as a "fuel switch" that has been shown to become an adaptive response to stress with the course of heart failurein diabetic and non-diabetic individuals.
In addition to acting as an alternate "fuel," beta-hydroxybutyrate is a signaling molecule that inhibits endogenous histone deacetylase (HDAC).
It may boost histone or lysine acetylation of other signaling molecules. Beta-hydroxybutyrate has been found to reduce reactive oxygen species generation and activate autophagy.
Furthermore, beta-hydroxybutyrate acts as an inflammasome inhibitor of the NLR family pyrin domain-containing protein 3 (Nlrp3), reducing Nlrp3-mediated inflammatory reactions.
Despite all of the positive benefits of beta-hydroxybutyrate, some research has revealed that long-term exposure to beta-hydroxybutyrate may be harmful.
Furthermore, not all methods of raising beta-hydroxybutyrate levels in the heart are equally helpful in treating heart failure.
The optimal timing and treatment options for delivering beta-hydroxybutyrate to treat cardiac disease are uncertain and have yet to be discovered.
Beta Hydroxybutyrate As Cardioprotective In Animals
The failing heart's myocardial has a fetal-like metabolic profile, with decreased fatty acid oxidation and increased glucose consumption. Increased ketone body usage may improve the prognosis of diabetic heart failure patients. Failure to employ ketones might hurt the prediction.
The enzymes succinyl-CoA:3-oxoacid-coA transferase (SCOT) and beta-hydroxybutyratebeta-hydroxybutyrate dehydrogenase 1 are necessary for the final oxidation of beta-hydroxybutyrate. Other mouse models have similarly indicated the necessity of ketone body use.
The inability of the heart to metabolize ketone bodies owing to SCOT loss resulted in increased left ventricular volume and lower ejection percent in a cardiomyocyte-specific SCOT knock-out animal model.
This was followed by an increase in reactive oxygen species (ROS) in the myocardium, mitochondrial damage, and disruption of myofilament ultrastructure. Ketone bodies are primarily formed in the liver due to fatty acid oxidation.
Beta-hydroxybutyrate may be oxidized in extrahepatic organs such as the heart after being delivered via the circulatory system. Fasting may lower the extent of myocardial infarctions and the development of premature ventricular complexes and reperfusion-induced ventricular arrhythmias.
The ketone body (beta-hydroxybutyrate) may boost autophagic flow in the myocardium, decrease ROS generation in the mitochondria, increase ATP production, reduce mitochondrial edema, and restore mitochondrial membrane potential.
In canine models of tachycardia-induced myopathy, infusion may remarkably retain systolic function and improve pathologic ventricular remodeling.
Mice unable to use beta-ohBenadryl hydrochloric acid (BAH) had worse cardiac remodeling, but ketogenic food improves left ventricular remodeling in TAC/MI mice.
More research is required to enhance the distribution of ketone bodies as a possible treatment for various cardiac disorders.
Beta Hydroxybutyrate As Cardioprotective In Humans
Ketone body metabolism and use in individuals with heart failure have been studied. The rate-limiting enzyme SCOT, which is required for ketone body use, rises in individuals with end-stage heart failure.
Exogenous 3-hydroxybutyrate (3-OHB) administration to patients with HFrEF improves hemodynamics through improving cardiac output. Comorbidity control is the focus of HFpEF therapy.
Intensive fasting-like diets for five days are safe and beneficial in lowering indicators and risk factors for aging and age-related illnesses, including cardiovascular disease.
Weight reduction, lower triglycerides, LDL cholesterol, blood glucose levels, and improved HDL cholesterol levels are among the processes. Lower blood glucose and IGF-1 levels contribute to the benefits in a randomized controlled human experiment with 100 participants.
Furthermore, the beta-hydroxybutyrate level rose dramatically, offering evidence of the possible significance of this hormone in the prevention of cardiovascular disease.
The potential of stressed hearts to switch to ketone bodies as an energy source is independent of the patient's diabetesstate. It is unclear if the increased cardiac ketone body usage is the cause or outcome of cardiac dysfunction.
Thus, whether exogenous ketosis induction is advantageous in all cardiovascular conditions warrants additional investigation. It is also challenging to distribute ketone bodies safely and accurately to correct aberrant metabolism.
More research is required to establish the link between beta-hydroxybutyrate and human cardiac disease and the appropriate time and dosage of ketone body preparations.
Mechanism Of Action
- Roles of Oxidation:Ketogenesis is essential for cardiac health, particularly during times of stress such as heart failure. Ketone bodies are produced in the liver, and the heart might utilize them as a modest energy source at first and then expand their use if the heart fails. Beta-hydroxybutyrate acts as an alternate fuel in failing hearts, supplying more ATP than glycolysis without enhancing cardiac efficiency. These oxidative functions of beta-hydroxybutyrate have recently been examined.
- Non-Oxidative Functions:Despite several studies demonstrating the cardioprotective benefits of beta-hydroxybutyrate and its oxidative activities, the precise underlying mechanisms of cardioprotection remain unknown. Elevated beta-hydroxybutyrate is a successful therapy for epilepsy and serves a neuroprotective function in neurodegenerative disorders such as Parkinson's disease and Alzheimer's disease. Beta-hydroxybutyrate's neuroprotective effects may be mediated through mitochondrial metabolic modulation, suppression of the mammalian target of the rapamycin (mTOR) pathway, and decrease of glutamatergic excitatory synaptic transmission. Furthermore, beta-hydroxybutyrate has been linked to cancer cell stemness by regulating cell proliferation and promoting important transcriptional factor expression. These capabilities imply that beta-hydroxybutyrate works not just as an alternative fuel but also as a signaling molecule.
Endogenous Beta Hydroxybutyrate Maintains Mitochondrial Homeostasis
Histone deacetylases (HDACs) control gene transcription by balancing histone acetyltransferase acetylation activity. Histone deacetylases alter the function of other proteins by modifying the lysine residue.
Activity is increased in heart failure, diabetic heart, and myocardial I/R damage models. Small molecule Histone deacetylase inhibition has been proposed as a possible treatment method for various illnesses.
Histone deacetylase inhibitors' cardioprotective effects have been linked to various mechanisms, including apoptosis, inflammation, metabolism, and autophagy.
According to one research, pretreatment with suberoylanilide hydroxamic acid (SAHA) protects murine fibrosarcoma L929 cells against TNF-induced necroptosis.
Autophagy is a lysosomal degradation system that is necessary to remove proteins and damaged organelles. Mitophagy is a kind of autophagy that is primarily responsible for the removal of damaged mitochondria by lysosomes.
Mitochondrial damage is a cause of oxidative stress, which may impair mitochondrial function. As a critical component of both normal and pathological metabolism, beta-hydroxybutyrate has a significant impact on a variety of mitochondrial processes.
Beta Hydroxybutyrate Inhibits NLRP3 Inflammasome
The Nlrp3 inflammasome is a critical innate immune sensor that regulates caspase-1 activity and the production of proinflammatory cytokines.
Suppression of this sensor may slow the course of atherosclerosis, diabetes, Alzheimer's disease, and other aging-related functional losses.
There is a decreased amount of circulating beta-hydroxybutyrate in high salt-fed hypertensive rats, but dietary supplementation of the precursor, 1,3-butanediol, recovers kidney function, resulting in virtually normal blood pressure.
Beta Hydroxybutyrate Is G Protein Receptor (GPR) Endogenous Ligand
Beta-hydroxybutyrate is an endogenous ligand for a member of the G protein-coupled receptor family that may have anti-atherosclerosis and anti-inflammatory properties.
Its particular binding may activate the HM74a/PUMA-G adipocyte-expressed G protein-coupled receptors, reducing lipolysis and regulating fatty acid supply and metabolism. This shows that hydroxycarboxylic acid receptor 2 activations might be a viable therapeutic target for illnesses like atherosclerosis.
The medication has been shown to reduce sympathetic outflow and heart rate by antagonizing G protein receptors41, preserving metabolic homeostasis. However, it is yet unknown whether it can protect hearts from I/R harm through G protein receptors.
Other Non-oxidative Functions Of Beta Hydroxybutyrate
To maintain metabolic balance, ketone bodies (beta-hydroxybutyrate) operate as histone deacetylase inhibitors, Nlrp3 inflammasome inhibitors, and G protein-coupled receptor ligands.
They may enhance cardiac cell excitation-contraction coupling under hypoxia at concentrations as low as 0.5 mM.
Lysine β-hydroxybutyrylation (Kbhb) is a post-translational modification of histone triggered by increasing ketone body levels and may promote gene transcription and cell proliferation.
Lysine β-hydroxybutyrylation (Kbhb) has been linked to the overexpression of genes in the starvation-responsive metabolic pathways. It can potentially become a novel target for cancer therapy by modulating p53 activity.
SGLT2 Inhibitors Increase Beta Hydroxybutyrate
The sodium-glucose cotransporter-2 inhibitors (SGLT2i) are diabetic drugs to manage blood glucose.
They target renal glucose reabsorption in an insulin-independent way, which may generate glycosuria of 60–90 g/day. SGLT1 has the greatest affinity for glucose in the gastrointestinal tract and is also implicated in renal and cardiac glucose management.
Empagliflozin may minimize the risk of cardiovascular events with a larger risk of amputation at the level of the toe or metatarsal. SGLT2i may produce better volume status, glucose-lowering, etc., in heart failure patients.
The particular processes of producing the cardiac benefits are not well characterized. Mildly raised ketone levels may boost fuel efficiency and minimize ROS generation, improving cardiac function and the prognosis of heart failure.
SGLT2i can decrease Nlrp3 inflammasome activation in isolated macrophages, which has also been demonstrated in diabetic patients with cardiovascular illnesses.
Beta Hydroxybutyrate Applications
Given the advantages of exogenous beta-hydroxybutyrate in patients with heart failure, numerous therapies for beta-hydroxybutyrate delivery have been developed.
In patients with HFrEF, beta-hydroxybutyrate infusion may enhance cardiac output and lower systemic vascular resistance dose-dependently while maintaining myocardial external energy efficiency. 1,3-butanediol, medium-chain triglyceride (MCT) supplementation, KEs, and ketone salts are all oral forms of beta-hydroxybutyrate.
KEs are the most promising treatments, with lower effective dosages and fewer adverse effects. KEs are often composed of multiple ketones or ketogenic precursors, and their breakdown products may be taken straight into the bloodstream as ketone bodies or their precursors.
Several clinical studies have evaluated the effects of KEs in healthy and cardiovascular disease circumstances. In healthy people, KEs may drop blood glucose, free fatty acid, triglyceride levels, plasma insulin, ghrelin, glucagon-like peptide-1, and peptide tyrosine levels.
More research is being conducted to determine the impact of KEs on pre-diabetic individuals.
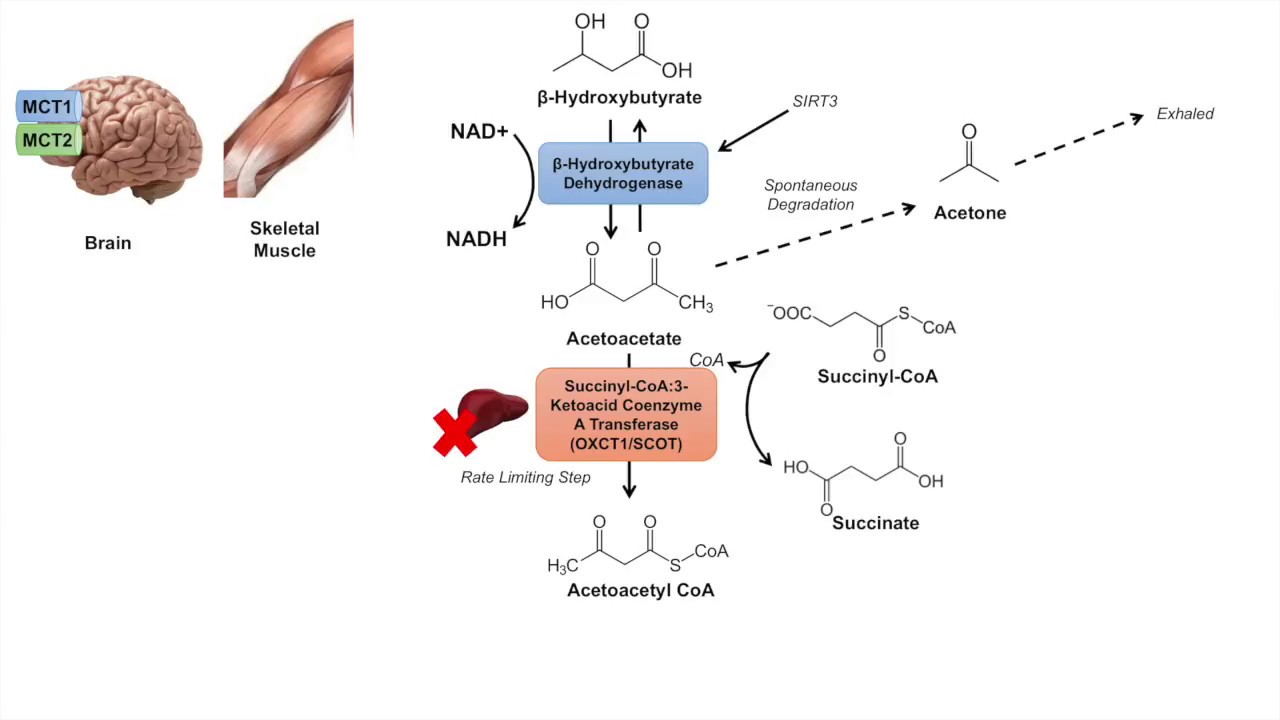
Ketone Body Metabolism | Ketolysis | Transport, Absorption and Catabolism
People Also Ask
Is Beta-Hydroxybutyrate A Ketone?
In mammals, beta-hydroxybutyrate is the most prevalent ketone body. Ketone bodies are tiny molecules circulating through circulation during fasting, prolonged activity, and carbohydrate restriction. They are predominantly generated in the liver from lipids.
Does Beta-Hydroxybutyrate Put You In Ketosis?
Acetoacetate and beta-hydroxybutyrate are the two primary ketone molecules generated during ketosis. Acetone is a less prevalent third ketone body. These ketone bodies supply energy to your brain, heart, and muscles by replacing glucose as fuel.
What Is A Natural Source Of Beta-Hydroxybutyrate?
Dairy milk includes amounts of beta-hydroxybutyrate ranging from 10 to 631 μM, making dairy products a natural source of beta-hydroxybutyrate.
What Does A High Level Of Beta-Hydroxybutyrate Mean?
Beta-hydroxybutyrate is a ketone body that is often seen in people with diabetic ketoacidosis. Testing for this ketone is crucial for those with type 1 and 2 diabetes.
Conclusion
Despite cardiovascular disease prevention, diagnosis, and treatment breakthroughs, they continue to be the primary cause of hospitalization and death.
Myocardial metabolism and enhanced use of beta-hydroxybutyrate under cardiac stressors such as heart failure and I/R damage have received particular attention.
There is some indication that long-term or high blood levels of beta-hydroxybutyrate are harmful to the heart.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles